
Dreamoar

Posts: 4711 Joined: 10-Sep-2009 Last visit: 27-Apr-2026 Location: Rocky mountain high
|
^ Was what you drank actually the "black" variety of Banisteriopsis caapi or was it Alicia anisopetala? The term "black ayhausca" has been used for both causing much confusion.
|
|
|
|
|

DMT-Nexus member
 
Posts: 12340 Joined: 12-Nov-2008 Last visit: 02-Apr-2023 Location: pacific
|
Vine and leaf wrote:According to my notes, it looks like I had the "honey caapi" pieces drying for six days before I started the soak process, so a week sounds totally fine there, Endless; it doesn't need to be cut the morning of the test or anything. I'm not sure if you looked at the other threads, but this now seems to have more to do with not only fresh vine, but this sweet sap that was being exuded after harvest. So I don't know if this is the case with all fresh vine, or only fresh vine which is exuding sap. I harvested the vine in late November, so I'm wanting to try cutting it the same time of year again, in case that had anything to do with it. If it does sap when I harvest it, and you're in the States at the time, I would be more than happy to send you some, if that's possible. I believe we may have a mutual friend who could get it to you if you're not comfy giving someone with one and a half posts here your address  . I've cut the same clone other times and not noticed the sap, but I haven't been looking for it, either. I'm really hoping that the next time I cut and dry, the stems will ooze the sap. That way, I could just scrape the sap and send it to someone for testing, along with a vine piece exuding the sap, a piece from a clone not exuding the sap, and a dried piece from the plant when it was exuding the sap last November, which I still have (again, no MDMA effect once its dried). But even more than that, I want this experience again, THIS WAS NOT HARMALA compound activity, or something that only potentates harmalas...this was a completely different chemical. And it didn't feel anything like a tryptamine, either. It was distinctly phenethylamine-like. Wow, never heard of anything like this before. I have never worked with fresh vine but minx has. Nice to see you here, EG. Long live the unwoke.
|
|
|
DMT-Nexus member
Posts: 22 Joined: 08-Apr-2012 Last visit: 25-Oct-2016 Location: FLA
|
Hey Jaime. I'm more of a voyeur over here. Hard to post most places anyway (a lot of wrong plant information I want to start arguments over, but can't; probably for the best as I waste enough time over at "church" as it is...)
So yesterday was peak full, and it's November, and my caapis are starting to yellow and lose some leaves as the days get shorter and cooler (we still haven't really hit near fifty degrees yet, but it's not eighty-three at four in the morning anymore) and so I harvested, just as the sun peaked up. Last year had been two-year old stems, two main ones twined around each other, thick as my fingers at a knuckle joint.
This year was one year old stems, most of them just as thick, but more than two, maybe five or six main stems. I cut them into manageable sized pieces and lay them down on the guest bed under the ceiling fan. All of the mumbo jumbo, sage and cannabis offering and such, remained the same as last year as well. I'm not exactly a scientist.
So, happier than a pig in shit to say, an hour or two after cutting the stems and laying them down, about half of them were oozing the honey. The "bottom" cuts were the ones initially doing it, so I am sticking with my assumption that the plant is preparing for less daylight by moving sugars to its roots through the phloem.
I don't think I should wait six days to brew, though. I think I should start the soak tonite. The thing is, the same vine, six months old, didn't have this effect and wasn't sweet, so whatever this is does oxidize, and so I think the sooner I can get to it with the hammer, the better. In fact, I'm about to.
I'll soak it for a week, brew for about a week, and then let everyone know if the brew is honey-sweet again or not. I'm trying not to get my hopes up, but man.
I managed to get a little bit of sap in a zip-loc, but barely any. I thought it would dry like a resin and I would be able to scrape it off, but it's too liquid, so instead it kind of just soaks into the wood of the stem around the cut, and makes these dark stains on the tips of the vine. So fail there.
|
|
|
DMT-Nexus member
Posts: 22 Joined: 08-Apr-2012 Last visit: 25-Oct-2016 Location: FLA
|
So I started the soak yesterday. The hammer, jars, handle on the jug of distilled white vinegar...everything was covered in a layer of stickiness, from the juices of the vine. The tile-top table that the vine pieces were resting on for an hour or two while I was pounding the daylights out of their brethren was covered in what looked like cat piss, was swarming with ants, and tasted (yeah, that's how I roll) like honey.
If I lick a piece of freshly cut vine, it's bitter. But if I let it sit for an hour or so, it will start oozing the honey (which is actually the consistency of water, not honey, but tasted just like honey. At least as sweet. I guess sugar cane juice would be a more suitable analogy, but probably less people are familiar.)
I will update again once the brew is reduced. Hopefully the final product will be as sweet as last year's, and as active.
Fingers crossed, y'all.
|
|
|
DMT-Nexus member
Posts: 22 Joined: 08-Apr-2012 Last visit: 25-Oct-2016 Location: FLA
|
Sorry, I'm basically just copying and pasting what I wrote over at the aya boards. To summarize, the sweetness is stronger, there is still a "different" effect going on, but so far it seems more centered in the head than the heart, but this head-centeredness may also be a result of heavy cannabis use the day leading up to the ceremony. I still felt "high" rather than "stoned," which is not typical of my cannabis/vine experience, so the "rush" effect was still very strong; it was just totally concentrated in my head; whereas my experiences with MDMA and mescaline (and the honey cappi last season) have all been undeniably focused on my heart. More bioassay is needed, and shall be done, probably this week.
********************************************************************************************
So.
I kept the cold-water soak separate from the boiled brew, and reduced both. I'm still not done brewing yet, but I have measurable, reduced amounts at this point, and man I could not wait, so I drank last night.
The boiled brew is normal. It is the color of black coffee when it's reduced and hot, then the color of slightly creamed coffee once cold. It has the consistency of creamed coffee; the same viscosity. The cold-soak, which was a two-day soak and a one-day soak, reduced, was bright yellow but with a blue tinge to it in the light, like an oil. It looked exactly like antifreeze. Once I started heating it started turning color, and by the time it was reducing it was the color of red wine. It also smelled exactly like red wine, minus the alcohol smell, while reducing. Now that it's reduced and in a jar, it looks like Chianti or grape juice, almost black but seems like a purple-maroon for the base color, until you hold it in front of a light and "roll" the jar back and forth, and see that it's actually a gold color. Instead of milky coffee, it has the consistency of very clean, very thin oil.
The boiled brew is normal. It is not sweet.
The cold-soak brew is even sweeter than last time. Five mL of it made 30mL of chacruna tea ridiculously sweet. It is like the sweetest bad wine (it tastes like wine, as well) that you have ever had in your life, followed very very quickly by the worst bitterestest alkaloid taste you have ever had in your life. Like, "uh, this is kind of weird/niceOHMYGODYECCHHHH."
The numbers first, then the report:
Total amount harvested/soaked: 2 Kg (two washes totaling 72 hours, 30mL distilled white vinegar added to each liter of distilled water used, room temperature, agitation five to ten times a day for thirty seconds.) I ended up with 550 mL. So let's call that 3.5g/mL. I drank thirty mL, which would be just over 100g cold-soak.
Total amount so far boiled: 1,200g (three washes of eight hours each, vine broken down after each wash, 30mL DWV for each liter distilled water used, brewed on high heat with lid on.) I ended up with 400mL. So let's call that 3g/mL. I drank 20mL, which would be 60g boiled.
So my dose, then is between 60 and 100 grams, fresh. More of the "goody oil" than a standard dose will be, unless I keep everything separate (and you know I will!) I still want to tinker with just the oil, but I wanted to try it all at once at first in case the thing in the oil needs an MAOi and the cold soak wasn't long enough to get any (I wonder if the oil has the sugar and the extra stuff, but isn't really ayahuasca, in other words).
So.
I feel tired and sluggish, and consider just going to bed. At 9:10 I drink both shots of caapi. 9:20 I feel something starting, but it's more in my head than in my heart. I smoke a bowl of c*nn*b*s and dance for a few minutes outside as Orion rises. I feel refreshed, invigorated, excited, like I'm at the airport going on vacation. The head is still the center of this energy, not the heart, but it's getting overwhelming. I go inside to sit down in front of the altar.
9:35 (25 minutes) I feel like I've taken 200mg of MDMA. Light closed-eye visuals, neck/base of skull pivoting, eyes rolling back and blinking in strange ways, bad nausea. I put some Santo Daime hymns on the headphones for a while, then some Deadmau5. The Deadmau5 kind of "peaked" whatever the rush-component is, and by 10:05 I feel like I'm back to baseline. So, once again, this "MDMA rush" feeling started rather early (by fifteen minutes) and then peaked and subsided at the hour mark. I can still "peak" at this point by focusing on my breathing, but I have to work on it. This intoxication did not feel like a typical ayahuasca intoxication, at all. Again, it feels like MDMA. I like listening to EDM during this phase, which usually bothers me when I drink (but it's all I listen to on cactus!) and the Daime hymns seem nasal and whiny, which is totally unlike my usual aya experience. Right around the "come back to baseline" phase, music becomes altogether intolerable, which is also typical for me. The rest of the night is silent.
10:45 (1 hour, 35 minutes) I drink my usual dose of 40 chacruna leaves (30mL of tea) which I combine with 5mL of the honey oil. The whole 35mL is made very sweet.
11:20 (2:10 after caapi, 35 minutes after chacruna) Purge. Sweet, wonderful purge!
11:40 (2:30 after caapi, 55 minutes after chacruna) The light hits, hard. I feel it slurp into my intestines, and am "gone in sixty seconds," to take Jees's phrase. Everything is pretty much the usual ayahuasca journey after that.
********************************************************************************************
Again, these numbers represent fresh material here, so this dose of let's say eighty grams, would be the equivalent to ten grams of dried vine. This was not only ridiculously potent on its own, without any light, but was more than enough to provide full MAOi for the light.
So I still have the "honey oil" and the "ayahuasca" separate, and am more than willing to send some off for testing. Lemme know.
|
|
|

DMT-Nexus member
Posts: 562 Joined: 20-Nov-2009 Last visit: 07-Jan-2023
|
Very informative thread. It is almost as if THH, the mysterious substance is alluding some forms of GC/MS testing, even baffling scientists perhaps? THH has a bad habit of turning up as harmine when fluorescent detection is not incorporated, those 2 extra hydrogen atoms are tricky. In the Heffter Institute study by Callaway (1) he found 170mg of harmine and 110mg of THH in a typical brew and small amount of harmaline (typically 10% of harmine level) used bi-weekly by the UDV. Now, about the only other testing done recently on caapi except for this thread was when I ran across a 'pure alkaloid caapi extract' on ecrater that was analyzed by Millis Scientific Labs to contain similar UDV-like levels of THH & confirmed the identity of all 3 alkaloids via MS tandem. I think parts of the report are still up if you search over there. All of this testing does not appear to be cheap and can costs hundreds of dollars to analyze just one sample. Thanks for taking the time to do this Endlessness, I've read this thread over and over many times, you are a master of this specialty. In (3) below, we see that 170mg harmine found in the UDV brew is a highly activating dose for admixture plants. This may have nothing to do with testing, but even when THH is converted from harmaline, a good portion of it (about 40%) goes "missing" somehow, even Shulgin was only able to get 70% yield using the best of materials, the yields are always sub-optimal, why is this? (1) Here was the reported levels of harmine, thh, and harmaline from UDV brew study (pg 2 of 12): "Scientific Investigation of Ayahuasca: A Review of Past & Current Research" by Mckenna, Ph.D, Callaway, Ph.D, Grog, M.D. http://www.heffter.org/d...ireview/01/chapter10.pdf(2) Here was the method Callaway used for testing, (pg 2 of 6): "Phytochemical Analyses of 32 Banisteriopsis Caapi and 36 Psychotria Viridis" by Callaway, Ph.D., Brito, M.D., Neves, M.D. http://www.vetmed.helsin...ntit/Callaway-plants.pdfQuote:High pressure liquid chromatography (HPLC) with fluorescence
detection . The chromatographic column packed with c-8 material
and the mobile phase has 20% methanol, 20% acetonitrile and 60%
0.1m ammonium acetate buffer, adjusted to ph 6.9 with acetic acid.
Individual signals in the chromatogram were verified according
to retention time and molecular weight by liquid chromatographic mass
spec (LC-MS) using a VG thermospray-plasma probe coupled to a
VG TRio-2 quadrople mass spec.
I would imagine modern methods of GC/MS testing are much improved over Callaway's day, technology is always advancing. (3) Moderate and Strong activating amounts of harmine (pg 250 of 639): " Pharmacotheon: Entheogenic drugs, their plant sources & history" by Jonathan Ott. Quote:For Experiment 12, I increased the qty of harmine base to 120mg, and
ingested this along with 35mg DMT free-base. By 45 minutes after ingestion,
it was obvious the dose was psychoptic, and I experienced a distinct DMT
effect building to a peak at 1:05 after ingestion and maintaining
a plateau until 1:50, with the effects largely dissipated by 3 hours
after ingestion. This experience was of comparable intensity to
EXPERIMENT 6 involving ingestion of a similar amount of DMT (30mg;
0.38mg/kg) with extract of 4g of harmel seeds, and we may conclude that
120mg of harmine (1.5mg/kg) will effect sufficient in vivo MAO-inhibition
to render DMT active orally with a longer duration and about half the potency
of inhaled DMT vapor.
To confirm these results, I decided to make another experiment with the
harmine/DMT combinations, slightly increasing the amounts of both compounds.
Accordingly, I prepared a capsule containing 160mg of harmine base;
2.0mg/kg) and 40mg DMT base (0.5mg/kg).
This capsule of 160mg harmine base and 40mg DMT base was ingested in
EXPERIMENT 13 and indeed provoked a proportionally stonger
DMT effect with the first effects felt in 20 minutes, building to
a peak by 1:30 after ingestion with a plateau until 2:40, and
clearly diminishing effects at 3:00 after ingestion. By 4:00 after
ingestion there were no effects, nor after-effects. All in all,
the experience was quite pleasant and similar to EXPERIMENT 3 in
Ecuador with about 50 leaves of DMT-containing Psychotria viridis
per dose. And, as we all know, harmaline is about twice as potent as harmine. You may remember me as 69Ron. I was suspended years ago for selling bunk products under false pretenses. I try to sneak back from time to time under different names, but unfortunately, the moderators of the DMT-Nexus are infinitely smarter than I am.
If you see me at the waterpark, please say hello. I'll be the delusional 50 something in the American flag Speedo, oiling up his monster guns while responding to imaginary requests for selfies from invisible teenage girls.
|
|
|

DMT-Nexus member
Posts: 562 Joined: 20-Nov-2009 Last visit: 07-Jan-2023
|
Concerning Jonathan Ott's experiments in the last post above, Jonathan Ott at the time did not have access to nature's THH, as they knew next to nothing about it back then nor it's crucial role in the Ayahuasca ceremony. Role of THH in caapi vine: Rue contains less than 1% tetrahydroharmine. It plays no role in rue. It is only found in very significant concentrations in caapi. It is the 2nd highest concentrated alkaloid found in caapi, right behind harmine, in some variety of caapi it is found in the same concentration as harmine. But in most variety, it is found in slightly less concentrations then harmine. 1) one of the strongest healing components in caapi, found to help stop drug abuse & violent behaviors, also up-regulates serotonin receptor sites, a very positive benefit, which stimulates the growth of new receptors. (Callaway) 2) doubles the half-life of admixture plant before it gets "zapped" by mitrochondria in the brain, allowing the experience to last longer. (theorized by D.Mckenna) 2) shuts down serotonin signaling (agonist) at the brain's 5-HT1a receptors, which make up more than 80% of all brain 5-ht receptors, this allows "real ayahuasca visions to be made possible" All the other natural oral entheogens do the same thing at the brain's 5-HT1a sites. (Nichols & subjective experience ie visions) 3) greatly reduces fear when taking ayahuasca (subjective experience) 4) increases focus and concentration, while decreasing confusion (subjective exp) 5) higher frequency vibration felt thru body (subjective exp) 6) increased appreciation of music, art, beauty & all things spiritual (subjective exp) 7) afterglow which lasts for a day or more (subjective experience) 8. has no abuse potential (subjective experience) 9) induces a strong tolerance of many weeks (subjective experience) 10) known as "the force" by shamans, vines high in THH are highly prized by them (Metzner) Also, subjectively, THH appeared to have some sort of visual 5-Ht2A or possibly adrenal agonism in order to balance out it's 5-ht1a agonism. How else to explain the glowing colors, sparkle & sacred look to everything? It also has strong effects on hearing. Recall that 5-ht1a agonism is inhibitory, while both 5-ht2a and adrenal agonism is stimulatory, yin and yang, so no matter what dosage of thh, it's effects were always balanced in full, nature is ingenious. It's too bad Thh was never analyzed for it's full complete receptor activity, I'm sure if it would have been, it would have come up a treasure trove of activity, see below for more: The Receptorome project by Thomas S. Ray: http://www.plosone.org/a...1%2Fjournal.pone.0009019hxxp://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0009019 You may remember me as 69Ron. I was suspended years ago for selling bunk products under false pretenses. I try to sneak back from time to time under different names, but unfortunately, the moderators of the DMT-Nexus are infinitely smarter than I am.
If you see me at the waterpark, please say hello. I'll be the delusional 50 something in the American flag Speedo, oiling up his monster guns while responding to imaginary requests for selfies from invisible teenage girls.
|
|
|

DMT-Nexus member
Posts: 562 Joined: 20-Nov-2009 Last visit: 07-Jan-2023
|
"Various alkaloid profiles in decoctions of Banisteriopsis caapi" Callaway, James C. (June 2005) http://catbull.com/alamu...in%20aya%20decoction.pdfIn the study above (see page 2) notice how some samples had more THH than harmine, others more harmine than THH. You may remember me as 69Ron. I was suspended years ago for selling bunk products under false pretenses. I try to sneak back from time to time under different names, but unfortunately, the moderators of the DMT-Nexus are infinitely smarter than I am.
If you see me at the waterpark, please say hello. I'll be the delusional 50 something in the American flag Speedo, oiling up his monster guns while responding to imaginary requests for selfies from invisible teenage girls.
|
|
|

DMT-Nexus member
Posts: 53 Joined: 07-Oct-2014 Last visit: 07-Nov-2018 Location: New Mexico
|
Just thought I would bump this topic. It's incredibly interesting, I hope to see more info. Open your mind and expand your conscious.
|
|
|

DMT-Nexus member
Posts: 4031 Joined: 28-Jun-2012 Last visit: 05-Mar-2024
|
Powdered Muricata, unreduced tea, 1 hour boil, harmine crystals after ammonia base. Jees attached the following image(s):  Muricata.1hrBoil.nonevap.25.5.17.jpg (172kb) downloaded 291 time(s).
|
|
|

DMT-Nexus member
Posts: 128 Joined: 03-Jun-2017 Last visit: 14-Jun-2022 Location: European Union
|
Here is a spreadsheet with all of the data in the first post so far. muladharma attached the following image(s):  Screenshot_20171011_124042.png (46kb) downloaded 267 time(s).
|
|
|
DMT-Nexus member
Posts: 3 Joined: 24-Oct-2018 Last visit: 26-Nov-2018 Location: Los Angeles, CA
|
endlessness wrote:This thread will grow as more analysis is gathered. Related Threads:Harmala and other less known B carbolines - Ongoing research thread with pharmacology, plant sources, chemical and physical data, analysis info, etc on different beta carbolines Poorly understood family of Ayahuasca vines (thread with plenty of info on different ayahuasca varieties, ayahuasca "colors" and unknown ayahuasca plants of the Banisteriopsis, Alicia and Tetrapterys genus, possible vendor misidentifications, etc) Conversion of Harmine->Harmaline->THH by simple boiling (and storage?)Literature:Schwarz et al 2003 Activities of extract and constituents of Banisteriopsis caapi relevant to parkinsonismKawanishi et al 1982 Shihunine and Dihydroshihunine From Banisteriopsis caapiCallaway 2005 Various alkaloid profiles in decoctions of Banisteriopsis caapiMckenna et al 1984. Monoamine oxidase inhibitors in South American hallucinogenic plants: Tryptamine and β-carboline constituents of AyahuascaHashimoto & Hashimoto 1976 New alkaloids from Banisteriopsis caapiSamoylenko et al 2010. Banisteriopsis caapi, a unique combination of MAO inhibitory and antioxidative constituents for the activities relevant to neurodegenerative disorders and Parkinson's diseaseHashimoto & Kawanishi 1975 New organic bases from amazonian Banisteriopsis caapi.Chemical profile described so far in literature:Harmine Harmaline THH Banistenoside A Banistenoside B Shihunine Dihydroshihunine Harmic acid methylester Harmalinic acid Harmic amide Acetyl norharmine Ketotetrahydronorharmine Tetrahydronorharmine β-d-fructofuranosyl-(2 → 5)-fructopyranose Nexus Analysis:GC-MSOther minor substances labelled in graphs still unidentified, work will be done in the future. Notice this is NOT quantitative analysis, just approx relative ratio of alkaloids in each plant, The total alkaloid yield has not yet been measured for most of these plants. Yellow caapi (Banisteriopsis caapi Yellow from k1w1b04ncaya) 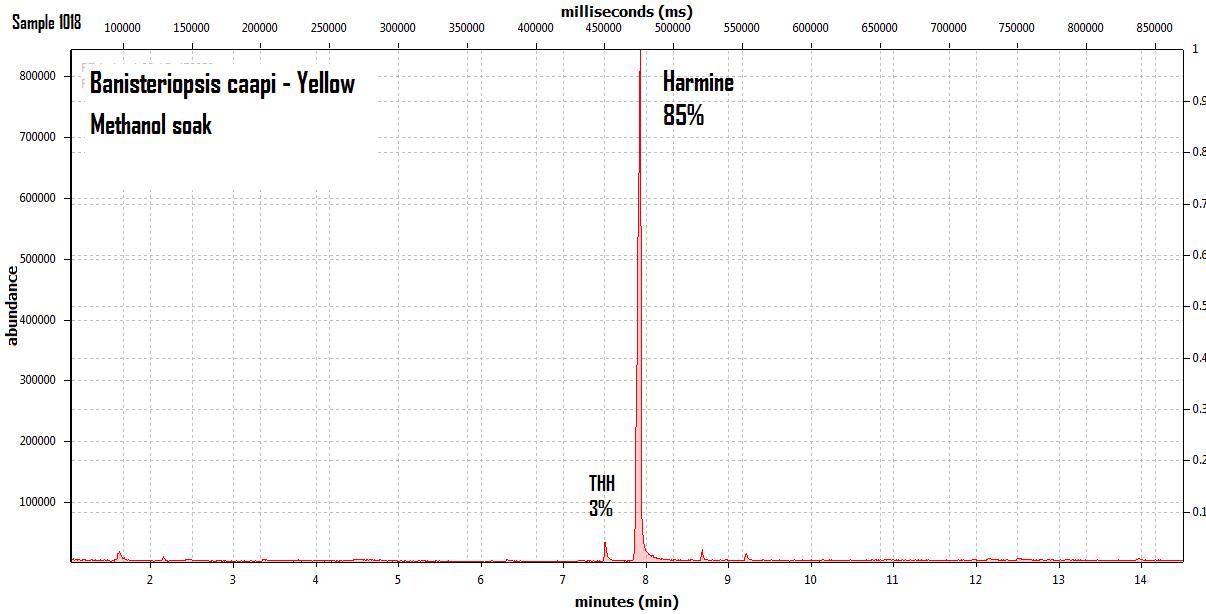 Ourinhos caapi Ourinhos caapi (Sold by IC4ROS-DN4) 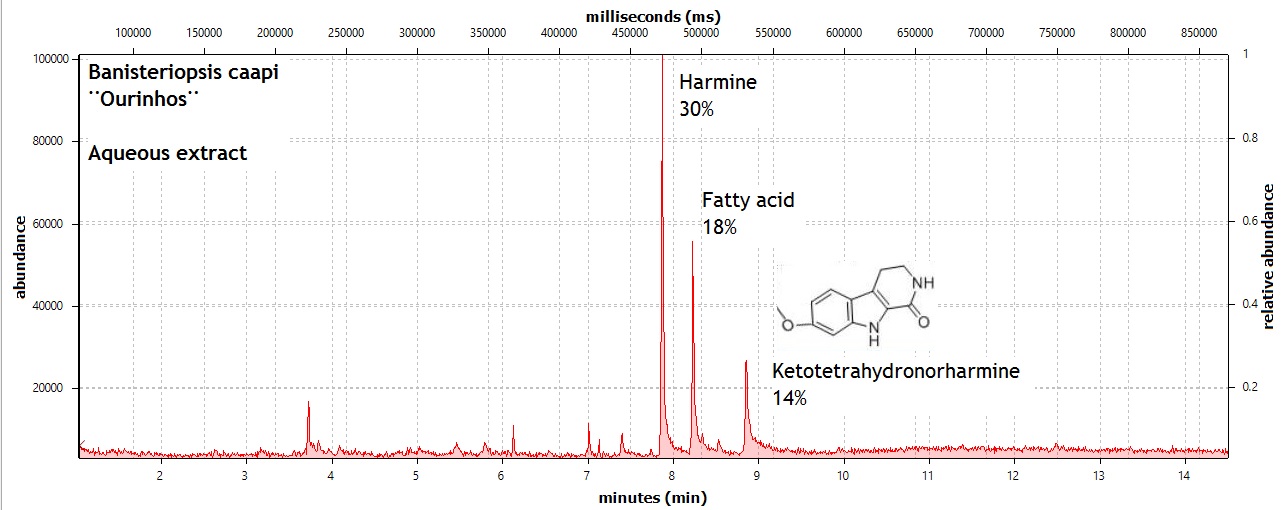 Red Ayahuasca Red Ayahuasca (Sold as Banisteriopsis muricata by k1w1b04ncaya) 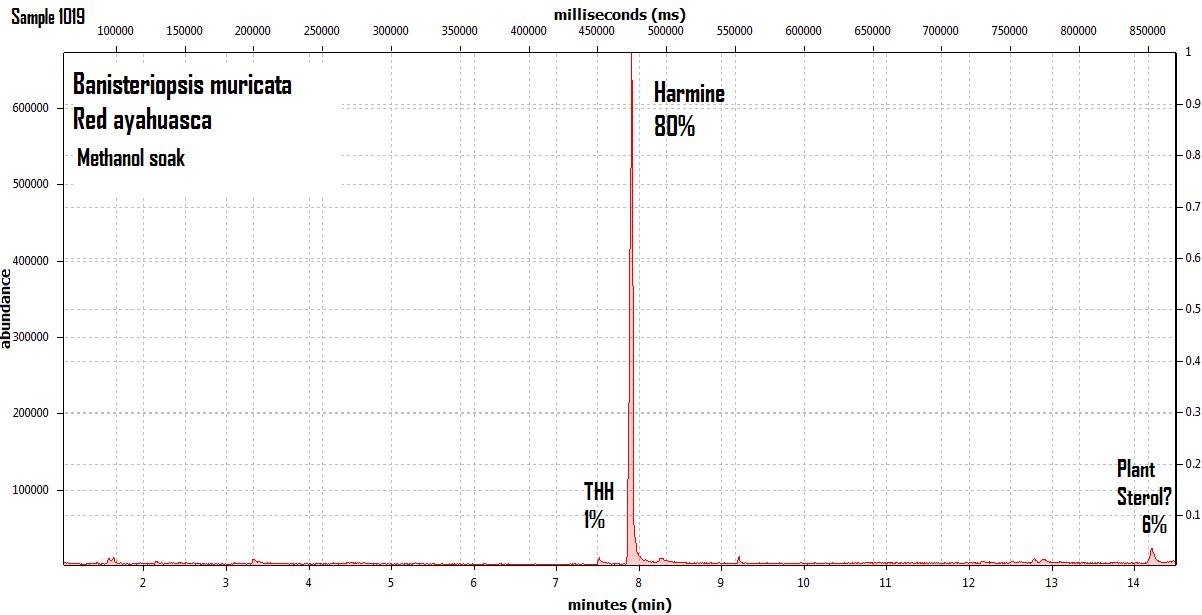 Red caapi Red caapi (from M4Y4 ethnobotanicals) Methanol soak 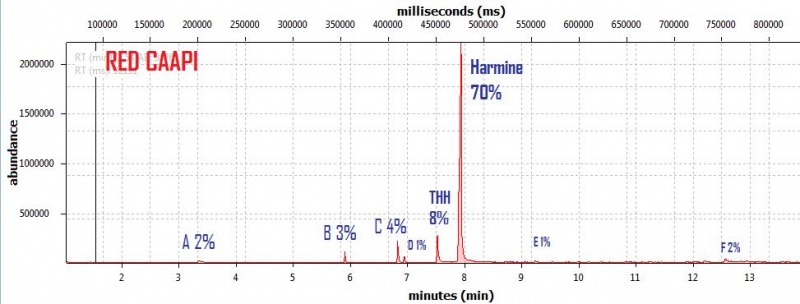 Black caapi NOTE: Some vendors have been selling what we identified as Alicia anisopetala as Black caapi/black ayahuasca. Note that A. anisopetala does NOT have harmala alkaloids as per my tests, pharmacology/toxicity is unknown. Please check this thread for more info. Black caapi NOTE: Some vendors have been selling what we identified as Alicia anisopetala as Black caapi/black ayahuasca. Note that A. anisopetala does NOT have harmala alkaloids as per my tests, pharmacology/toxicity is unknown. Please check this thread for more info.
(Tests of `black caapi` below are from M4Y4) (lets call this Black caapi #1) Methanol soak 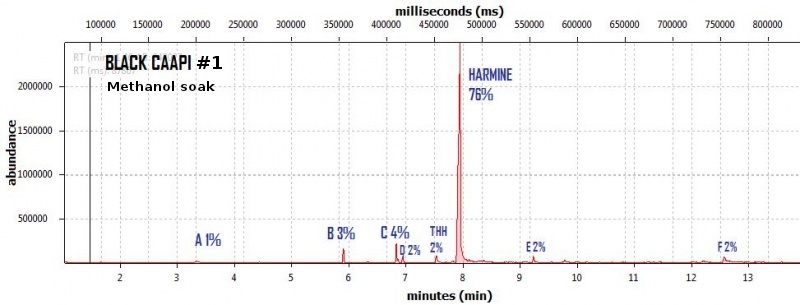 Black caapi Black caapi #1 A/B extract from same plant material as black #1 above. Yield 0.5% white powder.  Black caapi Black caapi #2 A/B extract, precipitation with NaOH. Yield 2.5%. From v3g3t4l1st4 botanicals. 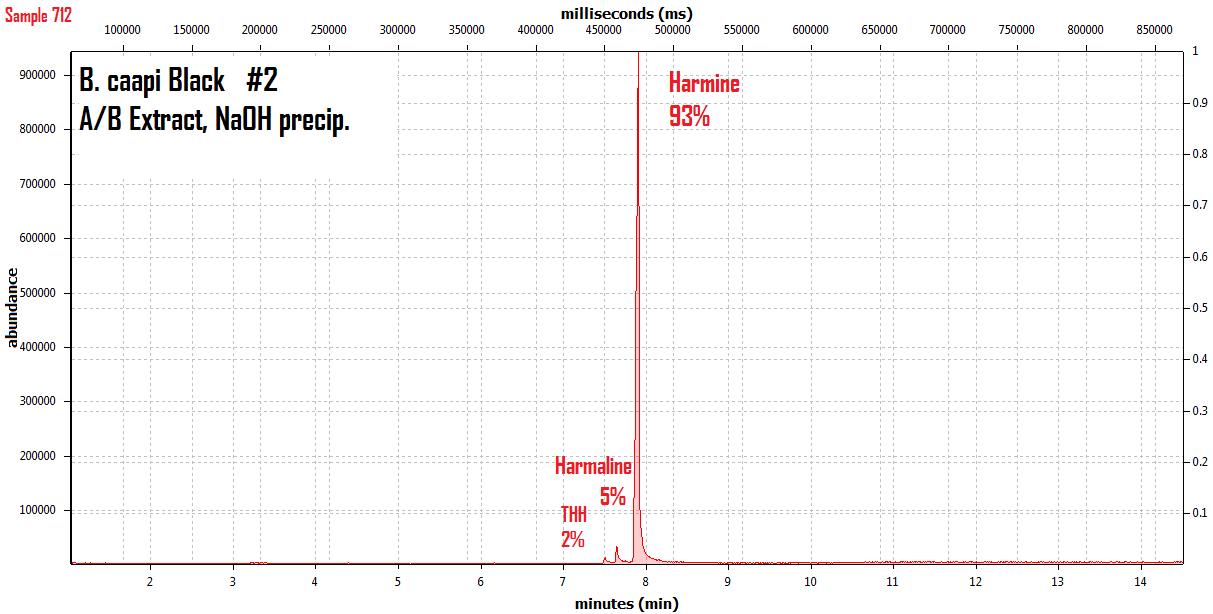 White caapi White caapi #1 Methanol soak (from MH---UK in nov 2011) A/B extract, precipitation with NaOH. 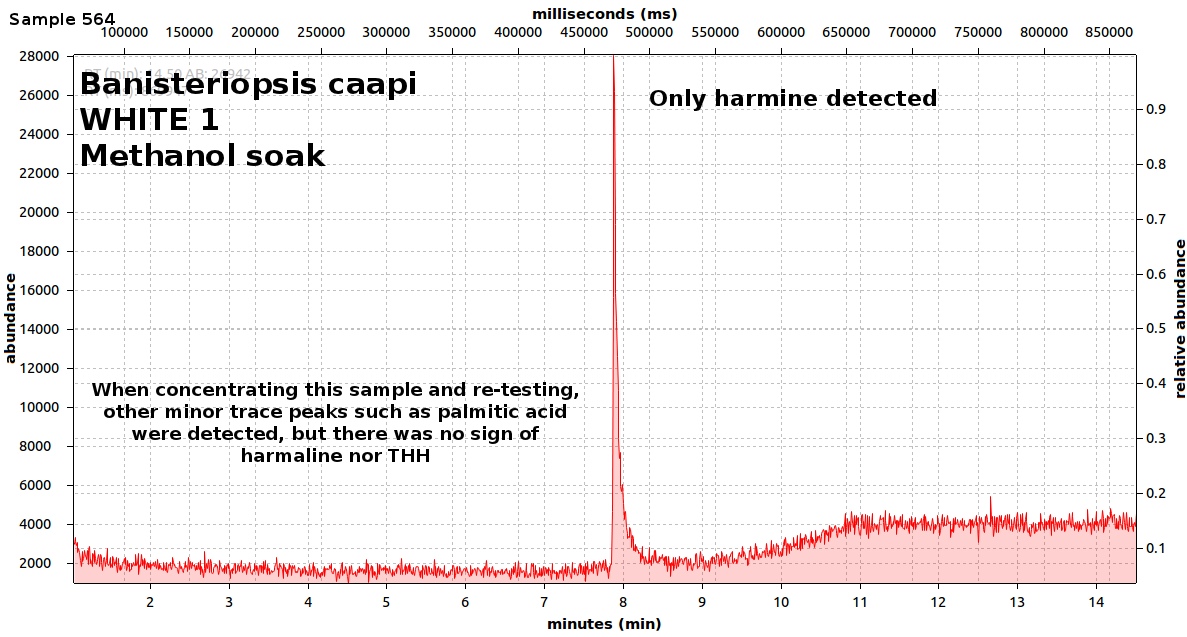 White caapi White caapi #1 A/B extract, precipitation with NaOH (from MH---UK in nov 2011) 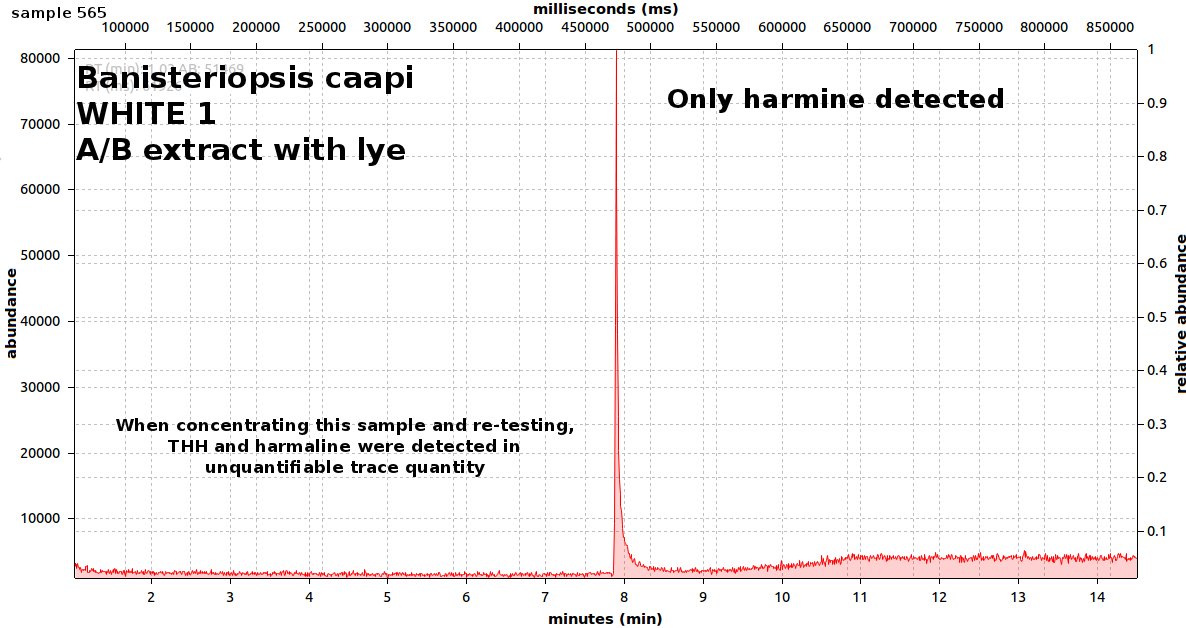 Bali caapi Bali caapi (was in freebies section of M4Y4) Methanol soak 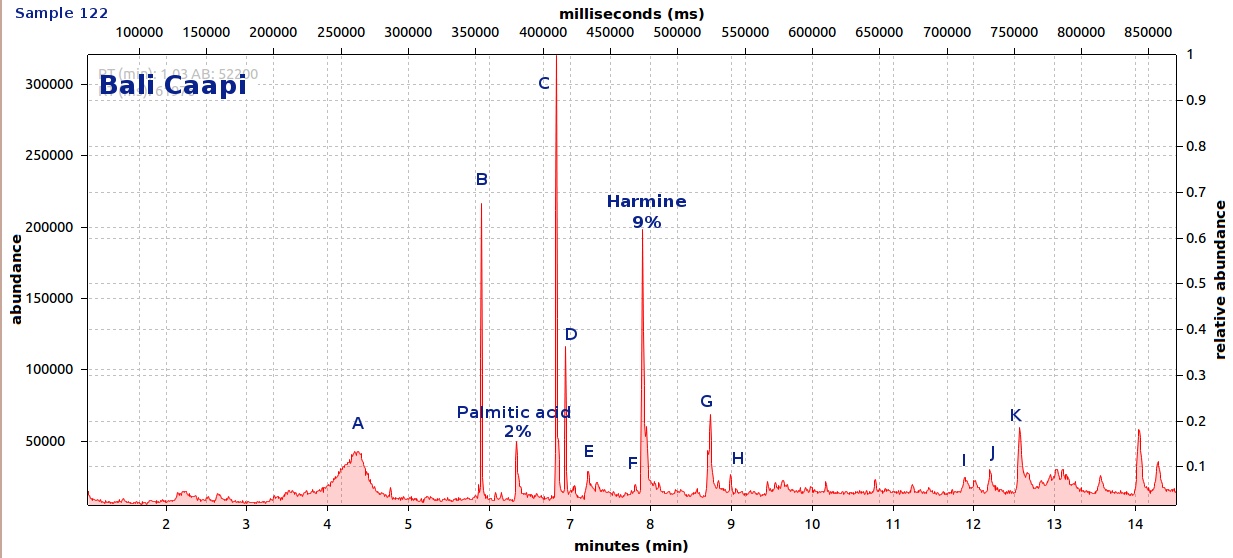 More caapi analysis to come in the future As always, im attaching the .MS file which you can open with openchrom to see the mass spectra (feel free to pm me if you want to learn how, its easy). Relative quantification here is approx, and subject to revision, bear with me im just starting with analysis. Here are the GC-MS details. In the future LC-MS analysis will also be done and info added below Quote:GC-MS analysis was performed in an Agilent 5890 series II gas chromatograph coupled to an 5971A quadrupole mass spectrometer detector (Agilent). The gas chromatograph was fitted with an 6890 auto sampler injector. Samples were injected in split mode into a 12m × 0.2mm i.d., 0.33 µm film thickness 5% phenylmethylsilicone column (ULTRA-2, Agilent Technologies). The oven temperature was programmed at 90ºC during 2min and increased 20ºC/min to 300ºC. Finally was maintained at 300ºC over 4min with a total run time of 14.50min. Insert liners packed with silanized glasswool were used. The injector and the interface were operated at 280 ◦C. Helium was used as carrier gas at a flow rate of 0.48ml/min. The mass spectrometer was operated in electron impact ionization mode at 70 eV
|
|
|
DMT-Nexus member
Posts: 3 Joined: 24-Oct-2018 Last visit: 26-Nov-2018 Location: Los Angeles, CA
|
endlessness wrote:This thread will grow as more analysis is gathered. Related Threads:Harmala and other less known B carbolines - Ongoing research thread with pharmacology, plant sources, chemical and physical data, analysis info, etc on different beta carbolines Poorly understood family of Ayahuasca vines (thread with plenty of info on different ayahuasca varieties, ayahuasca "colors" and unknown ayahuasca plants of the Banisteriopsis, Alicia and Tetrapterys genus, possible vendor misidentifications, etc) Conversion of Harmine->Harmaline->THH by simple boiling (and storage?)Literature:Schwarz et al 2003 Activities of extract and constituents of Banisteriopsis caapi relevant to parkinsonismKawanishi et al 1982 Shihunine and Dihydroshihunine From Banisteriopsis caapiCallaway 2005 Various alkaloid profiles in decoctions of Banisteriopsis caapiMckenna et al 1984. Monoamine oxidase inhibitors in South American hallucinogenic plants: Tryptamine and β-carboline constituents of AyahuascaHashimoto & Hashimoto 1976 New alkaloids from Banisteriopsis caapiSamoylenko et al 2010. Banisteriopsis caapi, a unique combination of MAO inhibitory and antioxidative constituents for the activities relevant to neurodegenerative disorders and Parkinson's diseaseHashimoto & Kawanishi 1975 New organic bases from amazonian Banisteriopsis caapi.Chemical profile described so far in literature:Harmine Harmaline THH Banistenoside A Banistenoside B Shihunine Dihydroshihunine Harmic acid methylester Harmalinic acid Harmic amide Acetyl norharmine Ketotetrahydronorharmine Tetrahydronorharmine β-d-fructofuranosyl-(2 → 5)-fructopyranose Nexus Analysis:GC-MSOther minor substances labelled in graphs still unidentified, work will be done in the future. Notice this is NOT quantitative analysis, just approx relative ratio of alkaloids in each plant, The total alkaloid yield has not yet been measured for most of these plants. Yellow caapi (Banisteriopsis caapi Yellow from k1w1b04ncaya)  Ourinhos caapi Ourinhos caapi (Sold by IC4ROS-DN4)  Red Ayahuasca Red Ayahuasca (Sold as Banisteriopsis muricata by k1w1b04ncaya)  Red caapi Red caapi (from M4Y4 ethnobotanicals) Methanol soak  Black caapi NOTE: Some vendors have been selling what we identified as Alicia anisopetala as Black caapi/black ayahuasca. Note that A. anisopetala does NOT have harmala alkaloids as per my tests, pharmacology/toxicity is unknown. Please check this thread for more info. Black caapi NOTE: Some vendors have been selling what we identified as Alicia anisopetala as Black caapi/black ayahuasca. Note that A. anisopetala does NOT have harmala alkaloids as per my tests, pharmacology/toxicity is unknown. Please check this thread for more info.
(Tests of `black caapi` below are from M4Y4) (lets call this Black caapi #1) Methanol soak  Black caapi Black caapi #1 A/B extract from same plant material as black #1 above. Yield 0.5% white powder.  Black caapi Black caapi #2 A/B extract, precipitation with NaOH. Yield 2.5%. From v3g3t4l1st4 botanicals.  White caapi White caapi #1 Methanol soak (from MH---UK in nov 2011) A/B extract, precipitation with NaOH.  White caapi White caapi #1 A/B extract, precipitation with NaOH (from MH---UK in nov 2011)  Bali caapi Bali caapi (was in freebies section of M4Y4) Methanol soak  More caapi analysis to come in the future As always, im attaching the .MS file which you can open with openchrom to see the mass spectra (feel free to pm me if you want to learn how, its easy). Relative quantification here is approx, and subject to revision, bear with me im just starting with analysis. Here are the GC-MS details. In the future LC-MS analysis will also be done and info added below Quote:GC-MS analysis was performed in an Agilent 5890 series II gas chromatograph coupled to an 5971A quadrupole mass spectrometer detector (Agilent). The gas chromatograph was fitted with an 6890 auto sampler injector. Samples were injected in split mode into a 12m × 0.2mm i.d., 0.33 µm film thickness 5% phenylmethylsilicone column (ULTRA-2, Agilent Technologies). The oven temperature was programmed at 90ºC during 2min and increased 20ºC/min to 300ºC. Finally was maintained at 300ºC over 4min with a total run time of 14.50min. Insert liners packed with silanized glasswool were used. The injector and the interface were operated at 280 ◦C. Helium was used as carrier gas at a flow rate of 0.48ml/min. The mass spectrometer was operated in electron impact ionization mode at 70 eV
|
|
|

DMT-Nexus member

Posts: 14191 Joined: 19-Feb-2008 Last visit: 22-Nov-2025 Location: Jungle
|
What happened there, did you just want to quote the original post? Was this an accident? Should I delete your posts?
|
|
|
pcdimitri
Posts: 1 Joined: 04-Jan-2019 Last visit: 22-Jan-2019
|
@endlessness Hey thats a great post thanks for the info on the caapi vine!
|
|
|

DMT-Nexus member
Posts: 1111 Joined: 18-Feb-2017 Last visit: 12-Jul-2024
|
I've posted my experience with a vine labeled B. muricata / red from Ecuador here. I'm curious as to its composition.
|
|
|

I rather root my values in my own hallucinations than in society´s neurotic illusions..
Posts: 681 Joined: 08-Jul-2017 Last visit: 08-Jul-2020 Location: Barcelona
|
https://www.dmt-nexus.me...;t=94521&find=unread"Too cute to live, too cozy to die" - Eaglepath
|
|
|

DMT-Nexus member
Posts: 1111 Joined: 18-Feb-2017 Last visit: 12-Jul-2024
|
A new paper with more data from dried vines and ready brews. Biodiversity of β-Carboline Profile of Banisteriopsis caapi and Ayahuasca, a Plant and a Brew with Neuropharmacological PotentialQuote:The objectives of this study were to investigate the chemical profile of B. caapi and of ayahuasca collected in various Brazilian regions. In total, 176 plant lianas, of which 159 B. caapi and 33 ayahuasca samples were analyzed. Dried liana samples were powdered, extracted with methanol, diluted, and analyzed by LC-MS/MS. Ayahuasca samples were diluted and analyzed. Mean concentrations in B. caapi were 4.79 mg/g harmine, 0.451 mg/g harmaline, and 2.18 mg/g tetrahydroharmine (THH), with a high variability among the samples (RSD from 78.9 to 170%). Native B. caapi samples showed significantly higher harmine concentrations than cultivated ones, and samples from the Federal District/Goiás had higher THH content than those collected in the State of Acre. The other Malpighiaceae samples did not contain β-carbolines, except for one D. pubipetala sample. Concentrations in ayahuasca samples ranged from 0.109 to 7.11 mg/mL harmine, 0.012 to 0.945 mg/mL harmaline, 0.09 to 3.05 mg/mL THH, and 0.10 to 3.12 mg/mL DMT. The analysis of paired ayahuasca/B. caapi confirmed that harmine is reduced to harmaline and to THH during the brew preparation. This is the largest study conducted with Malpighiaceae samples and showed a large variability in the main β-carbolines present in B. caapi.
|
|
|

DMT-Nexus member

Posts: 14191 Joined: 19-Feb-2008 Last visit: 22-Nov-2025 Location: Jungle
|
Thanks Jagube for sharing this paper, I hadn't seen it before.. I took a quick look at it and found a lot of interesting things being discussed. They looked at the difference of alkaloid content in different caapi "varieties", and didnt find a large difference. They also found more THH in caapi grown outside of the amazon in other parts of Brazil, which might have to do with environmental factors.
Regarding their conclusions on the reduction of harmine->maline->thh , it may be but I'm not entirely convinced on it. I'm wondering if THH in it's natural salt form is poorly soluble in methanol used for extraction of dried vines for analysis (while for the brewed vines they just diluted the samples to analyse) and hence the alkaloidal ratio difference. I think the way to actually test this is to compare caapi soak vs short brew vs long brew of same material.
|